Here's what it'll take to get a COVID-19 vaccine and how it'll be made

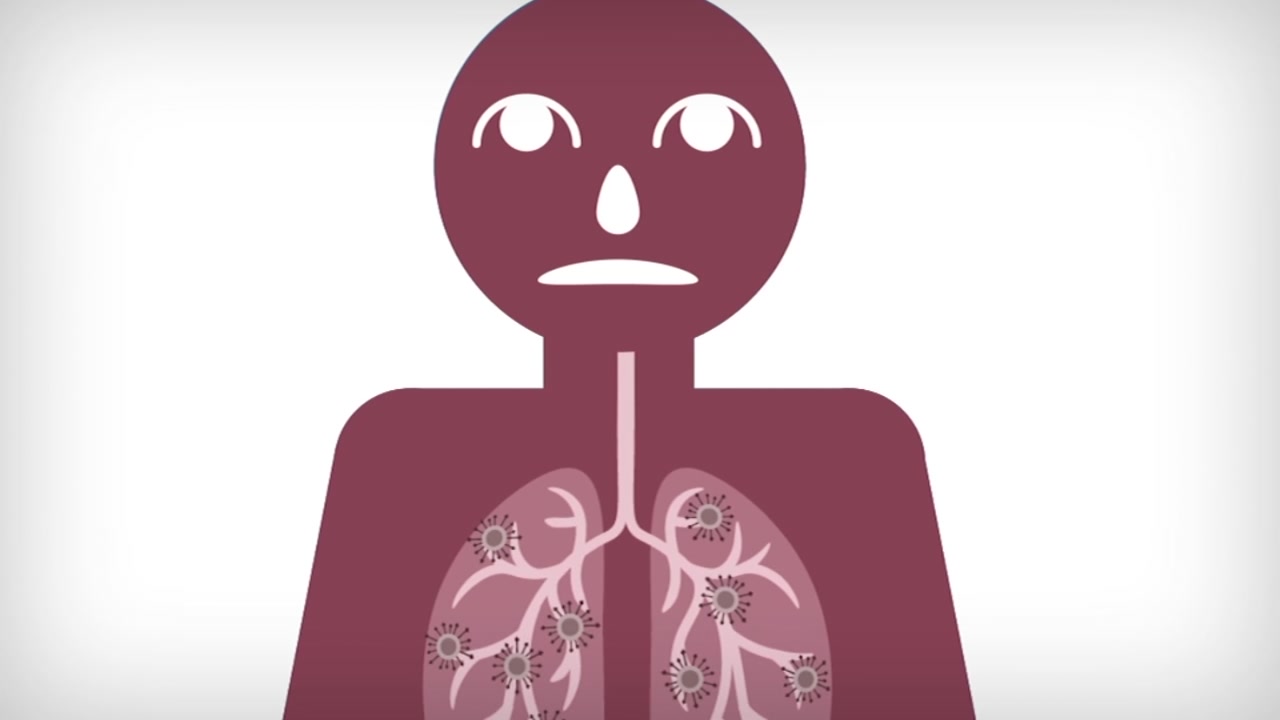
SAN FRANCISCO -- Novel coronavirus is likely going to be with us until a vaccine is developed. We know that COVID-19 attaches to your cells through your mouth and nose and works its way into your lungs where it can spread to other parts of your body. How do we stop it from doing that? With a vaccine. So what does it take to create one?
"A vaccine is a way for your immune system to identify a foreign invader and prepare for an attack even before you have an infection," said ABC7 News special correspondent Dr. Alok Patel.
A vaccine can stop a virus in its tracks, keeping you from getting sick.
"What scientists around the world are trying to do is create a vaccine that is going to teach your immune system what this coronavirus looks like so you are protected from a future infection," said Dr. Patel.
VIDEO: What does COVID-19 do to your body and why does it spread so easily?
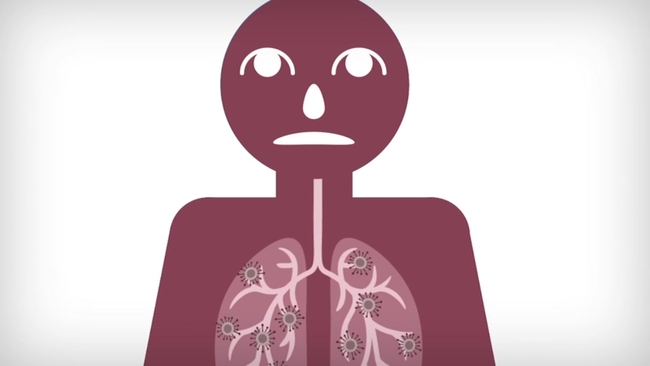
Vaccines are actually made from part of a bacteria or virus that have lost their ability to replicate and infect a person.
"This could be the outer shell of a bacteria of virus, it could be genetic material, it could even be a weakened version of it," said Dr. Patel.
That weakened version of the coronavirus can be injected into the body and recognized as a foreign invader, called an antigen. That antigen would then let your body know to protect it from the virus.
"What this is basically going to do, is give your immune system an idea of what the virus or bacteria look like," said Dr. Patel.
Before the first person is ever injected with a vaccine, it has to be proven in a lab first.
RELATED: Who will be first in line for a COVID-19 vaccine?
"Once they have that part down, and there's an idea that this type of vaccine could work, they then move on to phase one clinical trials," said Dr. Patel.
"Now phase one is going to use a very small amount of healthy adults, and make sure that the vaccine has no adverse side effects," said Dr. Patel. "You have to make sure that the vaccine is safe."
Phase two, would be to test the vaccine on hundreds of people.
At this point, researchers are looking at what the right dose is, and looking at how to scale up production of the vaccine to treat millions of people.
RELATED: '3rd wave' of COVID-19 likely in California, predicts top UCSF epidemiologist
In phase three, thousands of people would be involved in testing to make sure that vaccine is safe and works.
"Now this entire process, you guessed it, can take a couple of years," said Dr. Patel. "This is why, because after phase 3, all this work that these scientists have been doing gets reviewed by both the FDA and CDC."
The whole process typically takes 10 to 15 years before a vaccine can be licensed for use.
"So a lot of people now are saying, 'Whoa, 10 to 15 years that's a really long time,'" said Dr. Patel. "But everything changes when we are accelerating development because we are using previous research, and a lot more collaboration and a lot more funding. Which is why we are hopeful we could see a COVID-19 vaccine within 12 to 18 months."
With dozens of companies already testing out vaccines globally, researchers are hopeful the coronavirus may soon go the way of smallpox, malaria, and polio, and be part of "our" past.
VIDEO: Here's how shelter in place, stay at home orders can slow spread of COVID-19